Project Description
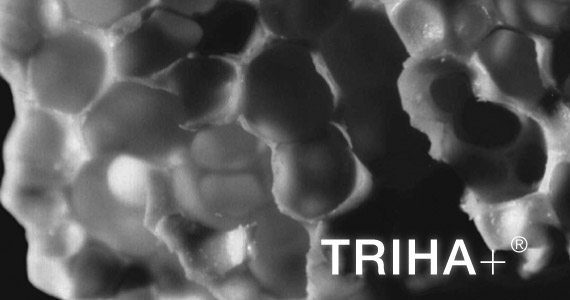
TRIHA+®
Synthetic Bone Substitute
TRIHA+® is a synthetic monophasic ceramic made of tricalcium phosphate (TCP).
Tricalcium phosphate Ca3(PO4)2 provides excellent osteointegration of the ceramic.
TRIHA+® is biocompatible and safe.
ADVANTAGES
- Biocompatible
- Synthetic
- Absorbable
- Osteoconductive
- Interconnected porosity
TRIHA+® is indicated for the filling of bone gaps or defects that are not intrinsic to the stability of the bone structure.
Example of use:
• Treatment of bone defects (benign tumors or cysts, traumatic lesions)
• Reconstruction during prosthetic revision
• Arthrodesis (foot, ankle, spine, …)
• Filling of osteotomy
• Filling cages
• Spine fusion
Monophasic ceramic 100 % TCP, an ideal answer to bone defects.
SYNTHETIC
• Free from organic phase
• No immunological risk
ABSORBABLE
• Minimum of 2 years
BIOCOMPATIBLE
• Compliant to ISO 10993-1**
**Biological Risk Assessment Report TRIHA+® 096/3/PERB
References
|
Designation |
References |
|
TRIHA+® 5 cc – 3x3x3 granules |
T824402 |
|
TRIHA+® 10 cc – 3x3x3 granules |
T824405 |
|
TRIHA+® 30 cc – 3x3x3 granules |
T824415 |
|
TRIHA+® 2.5 cc – 5x5x20 sticks (qty: 5) |
T827104 |
|
TRIHA+® 5 cc – 5x5x20 sticks (qty: 10) |
T827105 |
|
TRIHA+® 10 cc – Chips 1 – 2 mm |
T821210 |
|
TRIHA+® 20 cc – Chips 1 – 2 mm |
T821220 |
|
TRIHA+® 10 cc – Chips 2 – 5 mm |
T822510 |
|
TRIHA+® 20 cc – Chips 2 – 5 mm |
T822520 |